What is a photoinitiator?
photoinitiator, also known as (photosensitizer) or (photocuring agent),

01 Principle of Action
After absorbing light energy, photoinitiator molecules transition from the ground state to an excited state, undergoing chemical changes via singlet or triplet states to generate active fragments (such as free radicals and cations) that can initiate polymerization. Based on their mechanism, they can be categorized as follows:
– Free radical photoinitiators: These generate free radicals through cleavage or hydrogen abstraction reactions, initiating free radical polymerization.
– Cationic photoinitiators: After photolysis, they generate a strong proton acid (such as a Brønsted acid), initiating cationic polymerization.
02 Classification & Typical Representatives
Free Radical Photoinitiators:
(1) Cleavage-Type Initiators
After absorbing light energy, a Norrish I reaction occurs, and the bond breaks to generate free radicals:
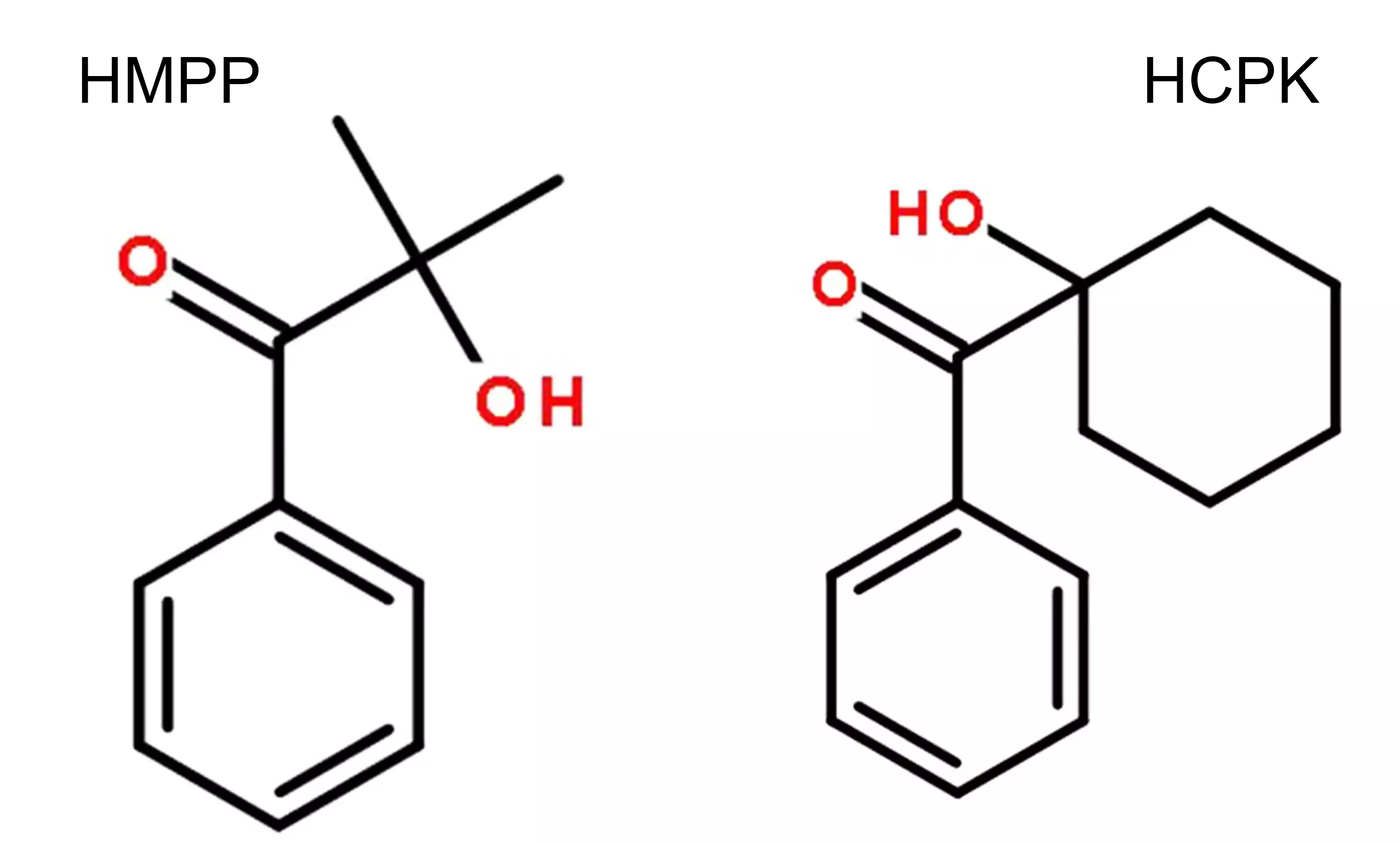
- 1173 (HMPP): 2-Hydroxy-2-methyl-1-phenylpropanone
- 184 (HCPK): 1-Hydroxycyclohexylphenyl ketone

659: 2-Hydroxy-2-methyl-1-[4-(2-hydroxyethoxy)phenyl]-1-propanone
MBF: Methyl benzoylformate
(2) Hydrogen abstraction initiators
Generate free radicals through hydrogen abstraction reaction:
- BP: Benzophenone
- MBP: 4-methylbenzophenone
(3) Composite initiator
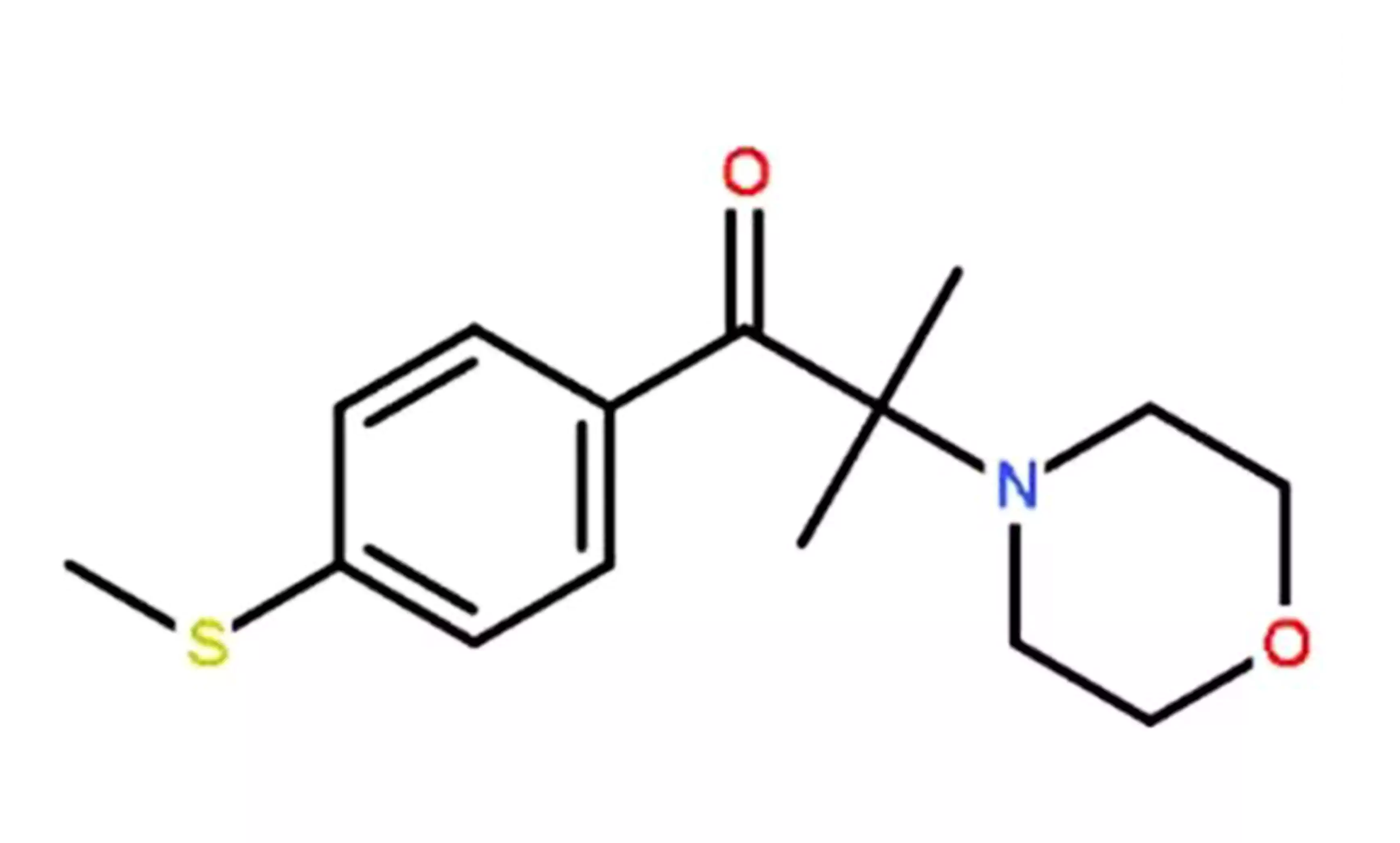
- IHT-PI 910:
2-dimethylamino-2-benzyl-1-[4-(4-morpholinyl)phenyl]-1-butanone
- IHT-PI 4265:50% TPO + 50% 1173
- IHT-PI 1000:20% 184 + 80% 1173
- IHT-PI 500:50% 184 + 50% BP
Cationic photoinitiators:
Photolysis generates a superacid that initiates polymerization of epoxies, vinyl ethers, and other materials:
- Ionium salts (e.g., I-250)
- Sulfonium salts (e.g., I-160)
- Metallocene salts (e.g., cumene ferrocenium hexafluorophosphate)
- Organosilanes
03 Structural Classification Summary
Free Radical Photoinitiators:
– Benzoin and Derivatives (Benzoin Ethers)
– Benzyls (Benzoyl Benzophenones)
– Alkyl Phenones (α-Hydroxyalkyl Phenones, α-Aminoalkyl Phenones)
– Acyl Phosphine Oxides (TPO, etc.)
– Benzophenones
– Thioxanthones
Cationic Photoinitiators:
– Onium Salts (Ionium Salts, Sulfonium Salts)
– Metalloorganics (Ferrocenium Salts)
– Organosilanes
04 Application Areas
Photoinitiators are core components of radiation curing technology and are widely used in:
UV coatings: wood, plastic, and metal coatings

UV adhesives: Electronics and medical adhesives
Gel polish: UV/LED-curable gel polish (such as CHROMÉCLAIR’s HEMA-free Gel Polish) for the nail art industry, offering fast curing, high gloss, and abrasion resistance
Other light-curing materials: 3D printing, photoresists, etc.
05 Features and Advantages
Light-curing technology meets the “5E” green standards:
- Efficient:Cures in seconds
- Enabling:Applicable to a variety of substrates
- Economical: Low energy consumption and high production efficiency
- Energy-saving: Room-temperature reaction, no heating required
- Environmentally Friendly: No VOC emissions, low pollution
